Recurrent connectivity and ribbon-like synaptic structures: how disease-vector mosquitoes sense carbon dioxide from human hosts
Mosquitoes. Their bite can be itchy, irritating - and potentially even lethal, given their role in spreading some of the world’s most serious infectious diseases [Ref 1]. Understanding mosquito physiology is a critical global health challenge, yet these dipteran insects remain considerably under-studied compared to their drosophilid relatives. When we think about mosquitoes, it’s mostly about how to avoid them. But turn that thought around, and we are left with an intriguing question:
How does the mosquito find you?
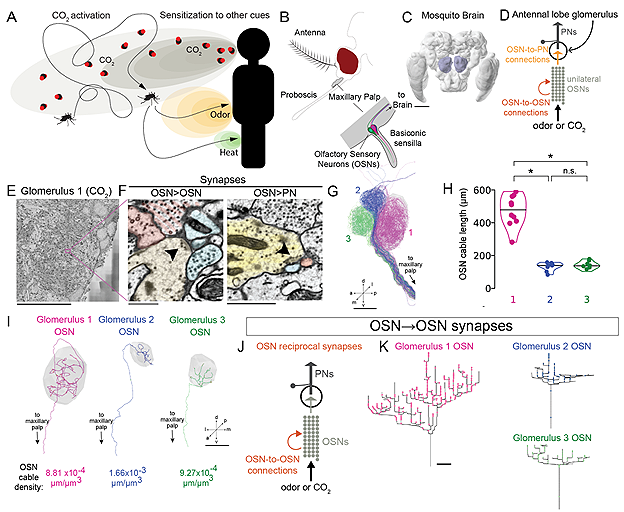
In fact, the mosquito diet is mostly based on plant sugars, with blood-drinking reserved for one specific purpose: laying eggs. Adult female Aedes aegypti mosquitoes use human blood as a protein source for egg maturation. To do so, they integrate a range of sensory cues, from body heat to odorant and visual signals [Ref 2]. One of the most critical host cues is carbon dioxide (CO2), which induces a behavioural state of active searching known as activation [Ref 3]. The anatomical basis of olfactory circuitry – which includes CO2 sensing – is similar in mosquitoes to that in drosophilids, but the specifics of neuronal morphology and synaptic connectivity remain unclear. How might these have diverged to reflect their different feeding strategies?
At Aelysia, we were delighted to contribute our ground truthing services to a fascinating new project from the Lee lab at Harvard Medical School and the Younger lab at Boston University, recently published as a preprint in BioRxiv [Ref 4]. In this project, Bao, Alford and colleagues considered whether the chemosensory coding strategies underlying CO2 sensitivity in Aedes aegypti are different to those of the model fruit fly Drosophila melanogaster. Using automated transmission electron microscopy (EM) of the A. aegpyti antennal lobe, they generated an EM volume that encompassed the three glomeruli innervated by the olfactory sensory neurons (OSNs) of the maxilliary palp, which mediates CO2-sensing in mosquitoes. This volume could then be directly compared to FlyWire, the recently completed D. melanogaster whole-brain connectome [Refs 5-6], which revealed striking inter-species differences.
In dipteran olfactory processing, OSNs converge on specific glomeruli based on expression of olfactory receptors, creating distinct local circuits with feed-forward and lateral connections. However, when reconstructing the A. aegypti maxilliary palp glomeruli, Bao, Alford and colleagues found that the connectivity of the CO2-sensitive glomerulus 1 was markedly different to that of the odorant-sensitive glomeruli 2 and 3. OSNs innervating glomerulus 1 had a remarkably high number of recurrent synapses - that is, synapses between two OSNs where both send axons to the same glomerulus - with 283 recurrent synapses per neuron, compared to 59 in glomerulus 2 and 69 in glomerulus 3. This result held even after controlling for variable neurite cable length between the three glomeruli, and when calculating recurrent connections as a fraction of total synapses. In fact, when annotating feedforward connections between OSNs and their downstream projection neurons (PNs), they found that CO2-sensitive OSNs had a greater number of recurrent than feedforward synapses, whereas the opposite was true for odorant-sensitive OSNs. As recurrent connectivity is frequently associated with signal amplification in many systems, this could represent a mechanism by which A. aegypti detects low concentrations of CO2 at long distances.
Is recurrent connectivity a general feature of dipteran CO2 sensing, or an adaptation specific to mosquitoes? To find out, Bao, Alford and colleagues compared glomerulus 1 circuitry in A. aegypti to olfactory glomeruli in D. melanogaster, including the CO2-sensitive "V" glomerulus. While feedforward synapses were similar in number and density between species, recurrent connections were vastly increased in A. aegypti, with 11 times more recurrent synapses in glomerulus 1 than in any glomerulus within the fly. Furthermore, the feedforward synapses in glomerulus 1 did not resemble the V glomerulus any more so than any other D. melanogaster glomerulus, suggesting that circuit architecture is not primarily driven by expression of conserved gustatory receptors. Instead, it is likely that the connectivity patterns underlying chemosensory processing in A. aegypti and D. melanogaster have evolved alongside divergent ecological niches and feeding strategies.
In addition to its high density of recurrent synapses, connectomic reconstruction of the A. aegypti CO2-sensitive glomerulus also revealed a surprising anatomical feature: putative ribbon-like synapses. Most insect neurons, including those in D. melanogaster, show characteristic "T-bar" presynaptic structures when viewed under electron microscopy. As expected, OSNs of all three A. aegypti glomeruli contained T-bars, but OSNs in glomerulus 1 also contained a smaller number of atypical presynaptic structures. These elongated, vesicle-lined structures were strongly reminiscent of mammalian ribbon synapses, which support sustained, graded transmitter release and have never previously been identified in insects. Could ribbon-like synapses play a role in CO2 sensing? For now, further ultrastructural and physiological studies will be required before we can determine whether these structures represent functional synapses. Nonetheless, these striking results demonstrate the power of volumetric EM approaches in understanding the diversity of neuronal circuitry.
Read the manuscript at BioRxiv here: https://doi.org/10.1101/2025.07.29.667487
References
T. Iwamura et al. (2020). Accelerating invasion potential of disease vector Aedes aegypti under climate change. Nature doi: 10.1038/s41467-020-16010-4
I. V. Coutinho-Abreu et al. (2021). Human attractive cues and mosquito host-seeking behavior. Trends in Parasitology doi: 10.1016/j.pt.2021.09.012
T. Dekker et al. (2005). Carbon dioxide instantly sensitizes female yellow fever mosquitoes to human skin odours. Journal of Experimental Biology doi: 10.1242/jeb.01736
J. Bao, W. Alford et al. (2025). Recurrent connectivity supports carbon dioxide sensitivity in Aedes aegypti mosquitoes. BioRxiv doi: 10.1101/2025.07.29.667487
Dorkenwald, S. et al. (2024): Neuronal wiring diagram of an adult brain. Nature doi:10.1038/s41586-024-07558-y
Schlegel, P. et al. (2024): Whole-brain annotation and multi-connectome cell typing of Drosophila. Nature doi:10.1038/s41586-024-07686-5